The Laboratory of Transgenic Models of Diseases combines utilization of cutting-edge genome-editing technologies and in-deep phenotyping analysis of an animal model to uncover novel genetic mechanisms for particular human diseases. We use the CRISPR/Cas9-based technique and various molecular-biology approaches to generate novel genetic mouse models or study the function of a particular gene in cells, ex vivo, or to deliver targeting or therapeutic vectors into the organism.
Our group has several areas of interest; all of them are connected via mouse models helping us to decipher either new biological roles that have not been described so far, or to understand pathologic gene variants in the development of a human disease.
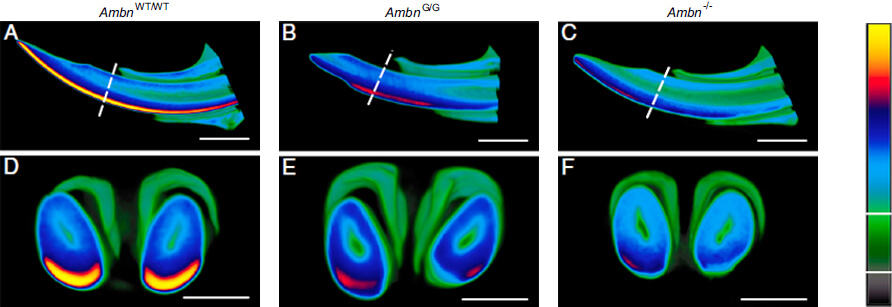
Thus, we study the role cullin-RING ubiquitin ligases involved in GIT homeostasis and pathological processes, since the cullin family has been largely associated with different types of cancer in GIT and thus represents a promising pharmacological target (Btbd3, Rnf121, Rnf186, Cul4a, Ddb1, Cul3, and others). In the field of proteases, we focus on studies of several metalloproteinases (e.g., Trabd2b), and especially on the functional redundancy of kallikreins and their inhibitors. If they are dysregulated (e.g., in the rare disease of Netherton syndrome), they have deleterious consequences in the disease development. In the field of craniofacial, skeleton and teeth development, we focus on the molecular mechanism driving the fascinating complex process of craniofacial and skeleton development, unveiling molecular regulation of epithelial morphogenesis and involvement of ubiquitin-dependent proteolytic pathways in the regulation of morphogenetic signalling cascades. One of the most striking features of craniofacial area is development of mineralized tissues such as teeth and bones. Here, we study the function of extracellular proteins, including ameloblastin, in the regulation of mineralization processes in tooth enamel formation and bone homeostasis process, revealing, for instance, how the FAM46A loss-of-function mutation was found in patients with osteogenesis imperfecta.
In 2020, in response to the pandemic situation based on Covid-19, we have developed several mouse models (Nature, 2021 May;593(7859):424-428) to study the biological aspects of SARS-Cov-2 infection in the mouse as a model organism and to develop therapies to this deleterious infection.